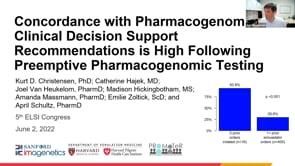
ELSIcon2022 Paper: Incorporating Patient and Relative Preferences in the Design of Traceback Cascade Testing Initiatives
ELSIcon2022 • Paper • June 3, 2022
Presented by: Katrina Romagnoli
Alanna Kulchak Rahm, Cabell Jonas, Tracey Klinger, Leigh Sheridan, Ilene Ladd, Zachary Salvati, Anna DiNucci, Paula Blasi, Aaron Scrol, Rachel Schwiter, Nora Henrikson
Background: People with ovarian cancer and their biological relatives can benefit from genetic testing for hereditary cancer risk, yet fewer than one-quarter of women with ovarian cancer are tested. A “traceback” cascade testing program seeks to retrospectively identify and offer testing to ovarian cancer survivors and their relatives. Feasibility and Assessment of a Cascade Traceback Screening program (FACTS) focuses on the feasibility of a traceback program with emphasis on communication strategies, the legal landscape, and an implementation science approach. We conducted a qualitative human-centered design interview study to understand the experiences and preferred communication strategies related to a traceback program.
Methods: We engaged ovarian cancer patients (n=31) and people with a family history of ovarian cancer (n=39) across a diverse sample from 3 U.S. integrated health systems. During video- and phone-based interactive interviews, participants designed their ideal experience of receiving communication about genetic testing. We conducted thematic and content data analysis using rapid framework analysis and affinity diagramming.
Results: A traceback genetic testing program was acceptable to participants. We identified 5 ideal experience phenotypes from passive to active outreach and personalized to generalized communication. We synthesized these results to develop preferred messages and design principles for traceback programs including: conducting personalized outreach with physician endorsement, providing access to clinician conversations, and using multiple modes of outreach.
Conclusions: These principles guide the integration of participants’ perspectives into the design of clinical traceback programs.
Tags
Videos in Series
-
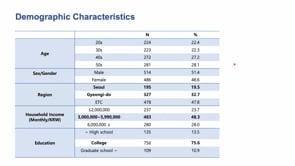
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: A Survey on Public Perception toward the Korean National Bio Big Data Project
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Extending the Belmont Principles: Sovereignty and Solidarity in Genomic Research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 12
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Public Engagement in Governance of Human Genome Editing in the Post-He Jiankui Era
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: "Extremely slow and capricious:" Genetic Researcher Experiences Using Shared Data Resources
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Consumers’ Perspectives Following Direct-to-Consumer Genetic Testing for Cancer Risk
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Flash Session 2
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Patients’ expectations of benefits from large-panel genomic tumor testing in rural community oncology practices
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Consumer Willingness to Share Personal Digital Information for Health-Related Uses
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Ensuring equity through insurance coverage for whole exome sequencing and reanalysis
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 14
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Moderated Plenary Panel
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Engaging Adolescents in Genomic Decision Making
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Facilitators and barriers to ethical machine learning in healthcare: A qualitative study on developer perspectives of potential harms
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 13
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Reproductive Concerns of Disabled Women: Whose Definitions of Disability are We Embracing?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: The Ethics of Influence and Choice Architecture in Genomics: Where Have We Been and Where Are We Going?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Disability Justice and Precision Medicine Across the Lifespan: Towards a Future of Equity and Access
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Ethical, Clinical, Legal, and Economic Issues Surrounding Genetic Variant Reinterpretation
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Neglected ELSI in Biobanking: Addressing Issues of Governance, Stewardship, and Equity
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Parent-reported barriers and facilitators to accessing care recommended from genomic sequencing in pediatric cancer patients
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Socioeconomic Status and Interest in Pursuing Genetic Testing for Hereditary Cancer in a US Based Population
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Democratizing Knowledge for Sickle Cell Disease Gene Therapy: A Deliberative Stakeholder Engagement Approach
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Embryoids: What are they, should we be concerned, should they be regulated, and if so, how?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 17
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Biological sample donation and informed consent for neurobiobanking: Evidence from a community survey in Ghana and Nigeria
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Fresh Air for Genetic Data Sharing
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 15
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Disrupting the status quo: a new model for benefit sharing in genomics research, a case study from South Africa
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Towards Justice- Race, ELSI, and Imagined Futures
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Provider Practices in Referring for Germline Genetic Testing for Men with Prostate Cancer at a Safety-Net Hospital
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Salt In My Soul: A Panel Discussion of the Documentary
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Is it just for a screening program to give people all the information they want?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: COVID, Prenatal Genetic Testing, and the Digital Divide: Beyond Technology
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: The Impact of Receipt of Genetic Test Results for Autism
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 10
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Meet the Experts - Trainee Session
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Cultivating just & equitable genomics research partnerships with community health centers
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 11
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Plenary: Troubled Dreams: Genetics, Race, and the Search for Justice in Sickle Cell Disease
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The Goldilocks Conundrum: Disclosing Discrimination Risks in Informed Consent
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 9
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Late-Breaking Abstracts, June 2, 2022
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Voluntary Genetic Testing in the Workplace: An ELSI Analysis
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Genetic data and the collective good: participants as leaders to reconcile individual and public interest
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Translating Genomic Science: How Genetic Counselors Navigate Ethical and Professional Dilemmas in the 21st Century
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Return of Secondary Genomic Findings: Experiences of Sickle Cell Disease Research Participants
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Participation in Sickle Cell Disease Clinical Research: How the Lived Experience Shapes Altruism
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Engagement with Historically Marginalized Communities: Methods, Positionality and ELSI in Precision Medicine Research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Why do Primary Care Patients Pursue Genetic Testing as an Elective Clinical Service? Findings from the Sanford Chip Program
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Navigating community engagement in clinical trial research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Plenary: Ableism, Audism, Ethics, and Genetics: A Just and Equitable Deaf Future?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Late-Breaking Abstracts, June 1, 2022
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Why Do Patients Drop Out of Population-Based Screening for Hereditary Cancer Risk?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 8
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Mapping Diversity and Equity in Precision Medicine Research: Towards an Ethics of Inclusion
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Ethical Topics Associated with Population Genomic Screening
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 7
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Mandating Diversity in the Context of Inequity: Re-examining the Research-Care Divide in Precision Medicine Research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: The Limits of Consent and Patterns of Coercion in U.S. Histories of Eugenic Sterilization
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: What Can Intersectionality Bring to ELSI?: Transforming Theory into Practice
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: How and why do researchers use “Ancestry”?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Interrogating the Value of Return: Stakeholders’ Perspectives on Return of Results in Biomedical Research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Sharing Qualitative Research Data: Addressing the Ethical and Logistical Challenges
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Permeable Membranes: How the blurring of boundaries and merging of silos should reshape ELSI research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Trainee Networking Session: Working with NHGRI
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Diversity’s Pandemic Distractions
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Exploring Disparities and Equity Across the Newborn Screening System
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Sharing Data: Speedbumps on the Translational Trail
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 6
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Late-Breaking: Victims of Eugenic Sterilization in Utah: Demographics and Survivor Estimate
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Clinician experiences and perspectives on the use of polygenic risk scores in child and adolescent psychiatry
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Development of a Consortium on Best Practices Related to Genomics and Adoption
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Seeking Diverse Inclusion in Bipolar Research: Lessons from a biobank
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 5
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Late-Breaking: South Africans’ opinions on human heritable genome editing: Promoting equal access as key to the future
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Child and adolescent psychiatrists’ knowledge, attitudes, and experiences with polygenic risk scores
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Rare Disease, Equity and Justice: Intersecting Disparities Across the Translational Spectrum
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The Genome and the Biome: Cystic Fibrosis @ Six Feet Apart
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 4
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Beyond the Binary: A Panel Discussion About Gender, Genomics, and Justice
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Listening Session: ELSI and the All of Us Research Program
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Communicating precision medicine research: multidisciplinary teams and diverse communities
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Patients’ and Parents’ Perceived Utility of Genomic Sequencing: Instrument Development and Validation
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 3
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Attitudes toward and actual uptake of genetic testing among a nationally representative sample of middle-to-older aged U.S. adults
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Cancer's Margins: Exploring Trans* Experiences of Hereditary Cancer Risk
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Genetic testing for hereditary cancer risk in India: A Case Study
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: How Did the Biodeterministic View of Criminality Influence Coercive Sterilization in the State of Iowa from 1934 to 1976?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 2
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: “Big bioethics” research: Is the ELSI community ready?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Late-Breaking Abstracts - May 31, 2022
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: An ELSI-informed strategy to use DNA data for humanitarian family reunifications
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: When State Laws Conflict: Choice of Law Challenges in Multi-Site Research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Paper Session 1
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: What Led to the Abolition of Iowa’s State Eugenics Program in 1977?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The Biologization of Morality: Criminal Justice as a Matter of Public Health
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022: Flash Session 1
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Using cost-utility analysis to evaluate the health equity impacts of population genetic screening for BRCA1/BRCA2
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Sociodemographic diversity in clinical genomics research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Precision medicine for whom? Upstream and downstream exclusion
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The Individual Genome as a Commons: Getting Genome Conceptualization and Governance Right
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Networking Session: Ethical Aspects of Reproductive Genetic Carrier Screening: Sharing Insights
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Welcome and Keynote: The Fallacy of Biological Race: Systemic Racism in Genomics
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Developing an Evidence Base for Investigative Genetic Genealogy (IGG) Policy-Making
-

The 5th ELSI Congress - ELSIcon2022 - ELIScon2022 Panel: Polygenic Embryo Screening: Ethical Considerations and Stakeholder Perspectives
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Informational Disparities in Electronic Health Records: Implications for Delivery of Hereditary Cancer Genetic Services
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Investigating an Ethos of Duty in Precision Medicine Research Calls for Minority Participation
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Self-Identified Spirituality and Interest in Genetic Testing for Hereditary Cancer Risk
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Genetic Attribution and Felt Stigma in People with Epilepsy
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Privacy, Agency, and Relationships in the Genomic Era: Views from the Public
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Community Perspectives on Electronic Consent Education for the Retention of Newborn Screening Bloodspots
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The Role of Patient Advocacy Groups Following Prenatal Diagnosis of a Genetic Condition
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Interest in genetic testing offered as part of routine healthcare among an ethnically diverse sample of young women
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Shining a light on overlooked questions in ELSI genomics
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Genetic Options and Constraints: How Genetic Ancestry Tests Change Ethnic Identities
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Race-Based Genetic Testing: Learning from Black Americans to Inform Policy and Practice
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Conceptualization and Measurement of Genetic Literacy
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The definition and handling of genomic data in a digitalized society: A Japanese perspective
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Latina women’s knowledge of, preferences for, and experiences with prenatal genetic testing: A scoping review
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: White racial framing and comfort with medical research: a critical analysis
-
The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Symbolic Legislation and the Regulation of Stroke Biobanking and Genomics Research in Sub-Saharan Africa
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: How does Genetics and Racial Disparities influence Stillbirth Risk and Reoccurrence?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Reconstruction of Normality Following the Diagnosis of Ehlers Danlos Syndromes
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Cystic Fibrosis Communities: Shifting Sites of Prognostic Imagination
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Panel: Out of the tower and onto the ground: engaging communities and scientists in emergent research priorities
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Systematic review of recommendations on the use of race, ethnicity and ancestry in the context of genetics research
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: How useful is genome sequencing to parents of pediatric cancer patients? Findings from the Texas KidsCanSeq Study
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: ELSI and the New Precision Stewards?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Reporting “Clinical Utility” in NICU Sequencing Studies: Systematic Review and Call for Rethinking
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Examining Embeddedness: A Call for Principles and Best Practices for Integrating ELSI
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Toward an anthropology of rare disease
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Physician Perspectives on Polygenic Risk Scores
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The 'Lived Experience' of Pediatric Gene Therapy - A Scoping Review
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: In the Shadows of Virtual Family Trees
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Resisting an old vice, or how polygenic risk information can do the most good
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Exploitation in Genetics Research: Vulnerability, Unfairness, and Degradation
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: What I learned about sequencing from my critically ill daughter
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: The ELSI of Pandemic Biobanking
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Inequality and Health: Sickle Cell Disease and Food Insecurity during the COVID-19 Pandemic
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: Revisiting the ethics of secondary findings in diagnostic genome-wide sequencing
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Family Secrets Revealed: Experiences and impacts of participating in direct-to-consumer genetic relative-finder services
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Whose future should we worry about?
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Paper: Population Frequencies of Hereditary Cancer Gene Variants in Diverse Population Groups
-

The 5th ELSI Congress - ELSIcon2022 - ELSIcon2022 Flash: The Perspective on Healthcare Providers on the Return of Secondary Genetic Findings in Africa